Posts
Comparison of laboratory-based and non-laboratory-based WHO cardiovascular disease risk charts: a population-based study
/0 Comments/in PCS Publications /by PCSAbstract
Background: Determining the risk of Cardiovascular Disease (CVD) is a necessity for timely preventive interventions
in high-risk groups. However, laboratory testing may be impractical in countries with limited resources. This study
aimed at comparison and assessment of the agreement between laboratory-based and non-laboratory-based WHO
risk charts models.
Methods: This study was performed using the baseline data of 8138 participants in the pars cohort study who had
no history of CVD and stroke. The updated 2019 WHO model was used to determine the 10-year fatal and non-fatal
CVD risks. In general, there are two types of new WHO risk prediction models for CVD. The scores were determined
based on age, sex, smoking status, diabetes, Systolic Blood Pressure (SBP), and total cholesterol for the laboratory-
based model and age, sex, smoking status, SBP, and Body Mass Index (BMI) for the non-laboratory-based model. The
agreement of these two models was determined via kappa statistics for the classified risk (low: < 10%, moderate:
10–< 20%, high: ≥ 20%). Correlation coefficients (r) and scatter plots was used for correlation between scores.
Results: The results revealed very strong correlation coefficients for all sex and age groups (r = 0.84 for
males < 60 years old, 0.93 for males ≥ 60 years old, 0.85 for females < 60 years old, and 0.88 for females ≥ 60 years old).
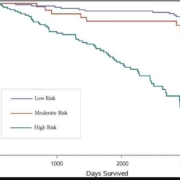
In the laboratory-based model, low, moderate, and high risks were 76.10%, 18.17%, and 5.73%, respectively. These
measures were respectively obtained as 77.00%, 18.08%, and 4.92% in the non-laboratory-based model. Based on risk
classification, the agreement was substantial for males < 60 years old and for both males and females aged ≥ 60 years
(kappa values: 0.79 for males < 60 years old, 0.65 for males ≥ 60 years old, and 0.66 for females ≥ 60 years old) and
moderate for females < 60 years old (kappa = 0.46).
Conclusions: The non-laboratory-based risk prediction model, which is simple, inexpensive, and non-invasive, classi-
fies individuals almost identically to the laboratory-based model. Therefore, in countries with limited resources, these
two models can be used interchangeably.
Agreement between laboratory‑based and non‑laboratory‑based Framingham risk score in Southern Iran
/0 Comments/in PCS NEws, PCS Publications /by PCSAuthors
Fatemeh Rezaei1 , Mozhgan Seif2 , AbdullahGandomkar3 , Mohammad Reza Fattahi4 & Jafar Hasanzadeh5
Abstract
Introduction: Estimation of the risk of cardiovascular diseases (CVD), may lead to prophylactic therapies. This study aims to compare and evaluate the agreement between CVD prediction of Iran Package of Essential Non-communicable Disease (IraPEN) and Framingham risk score (FRS).
Methods: All 40-79 years old participants in the Yazd Health Study who did not have a history of CVD were included. The 10-years risk of CVD was estimated by the laboratory (IraPEN), non-laboratory WHO-EMR B and FRS. The risk was classified into low, moderate and high-risk groups. Cohen’s weighted kappa statistics were used to assess agreement between tools. To assess discrepancies McNemar’s χ2 test for paired data was used. P values < 0.05 were considered statistically significant.
Results: In total, 2103 participant was included and the risk scores were calculated. Of them, 26.5% were stratified as high risk by FRS, compared with 6.1% by IraPEN. A slight agreement (37.9%) was observed (kappa 0.17, P < 0.0001), in other words. This discrepancy between IraPEN vs. FRS was seen in both sexes (P < 0.0001), although in women the agreement ratio was higher (52.1% vs. 21.3%). The discrepancy between FRS and IraPEN in categorizing people at risk of CVD was 55.5%, (P < 0.0001) but this was not significant between IraPEN and non-laboratory WHO-EMR-B (World Health Organization – Eastern Mediterranean Regional-B group countries) score (P < 0.523; discrepancies, 5.8%).
Conclusion: Our study shows a slight agreement between various CVD risk scores. Thus, reviewing the IraPEN and using alternative tools for the low-risk group should be considered by decision-makers. It is important to use a more reliable score for nation-wide risk assessment.
Developing Non-Laboratory Cardiovascular Risk Assessment Charts and Validating Laboratory and Non-Laboratory-Based Models
/0 Comments/in ICS Publications /by ICSContact
No.24, Aarabi St, Yaman St, Velenjak, Research Institute for Endocrine Sciences, Shahid Beheshti University of Medical Sciences, Tehran, Iran. Postal Code: 1985717413, P.O Box: 19395-4763
- Tel: +982122432500 (612, 457)
- Tel: +982122402463
- Fax: +982122416264
Latest News
 Webinar: Shahrekord Prospective Epidemiological Research Studies in Iran (PERSIAN) experienceJune 3, 2024 - 4:22 pm
Webinar: Shahrekord Prospective Epidemiological Research Studies in Iran (PERSIAN) experienceJune 3, 2024 - 4:22 pm Association between latent profile of dietary intake and cardiovascular diseases (CVDs): Results from Fasa Adults Cohort Study (FACS)October 18, 2023 - 11:43 am
Association between latent profile of dietary intake and cardiovascular diseases (CVDs): Results from Fasa Adults Cohort Study (FACS)October 18, 2023 - 11:43 am Association between adherence to the Mediterranean diet with cardiometabolic risk factors: a cross-sectional study on PERSIAN cohort study in FasaSeptember 8, 2023 - 8:09 am
Association between adherence to the Mediterranean diet with cardiometabolic risk factors: a cross-sectional study on PERSIAN cohort study in FasaSeptember 8, 2023 - 8:09 am